|
7/24/2023 0 Comments Lithium carbon dioxide formula
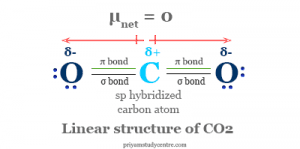
The overall knowledge and encyclopedia of Lithium carbonate (Li2CO3) covering characteristics, safety, usage. i) What type of reaction is this (1 mark) ii) Write a balanced equation for this reaction. Get to learn about Lithium carbonate (Li2CO3). most often caused by the formation of non-flammable carbon dioxide (CO2) gas. The products are solid lithium oxide and carbon dioxide. 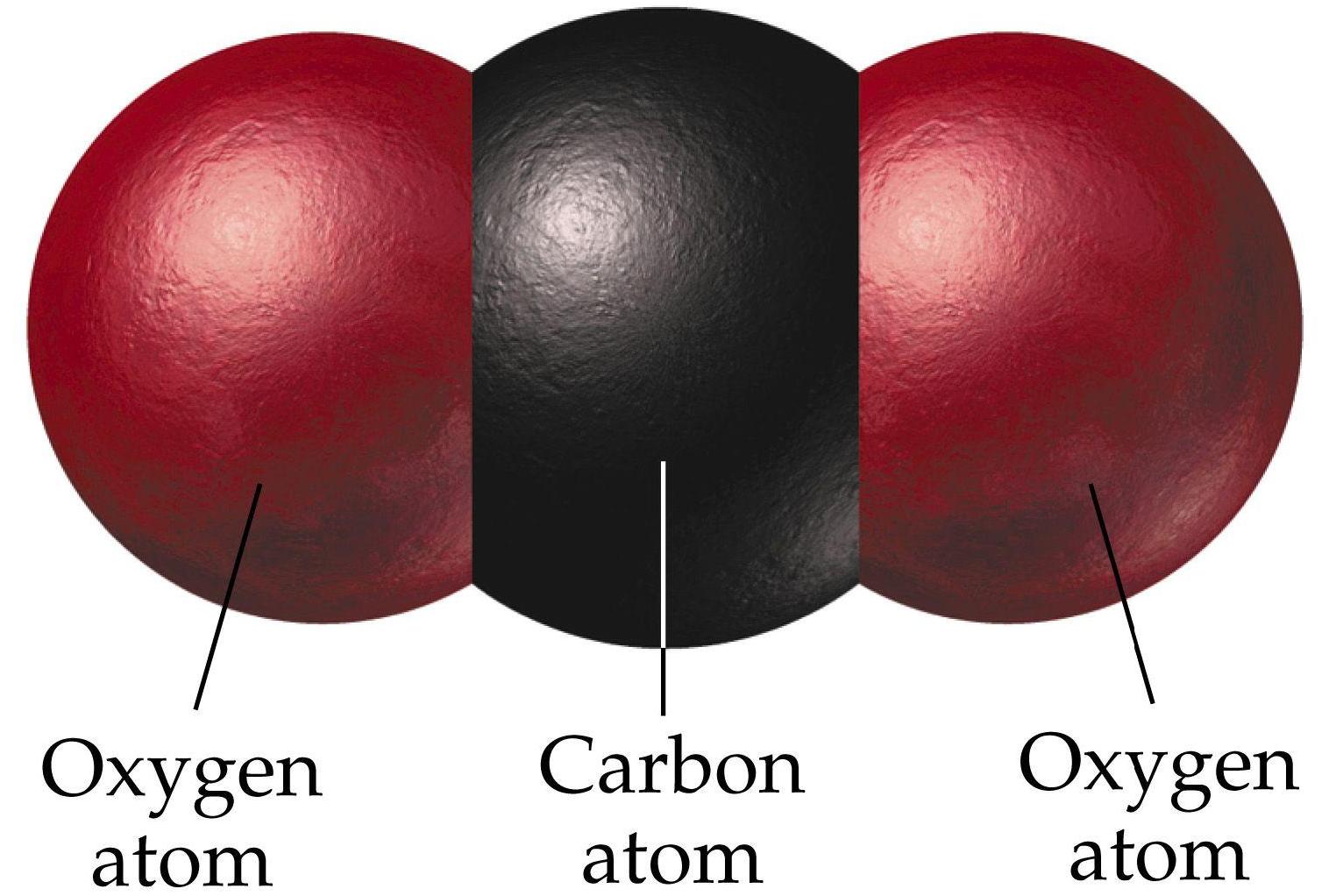
Lithium oxide reacts with water, by forming lithium hydroxide, Li 2 O + H 2 O 2LiOH. Lithium oxide can form lithium carbonate by absorbing carbon dioxide, Li 2 O + CO 2 Li 2 CO 2. Take a look at the balanced chemical equation for this decomposition reaction.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
